RESPOKARE N95 Mask RK-200-3040A/3041A/3042A N95 Mask Details
The Company began research efforts in 2007, culminating in the sale in 2013 of a surgical mask with an antimicrobial/antiviral agent that comes under the new FDA product code OUK. This mask is the genesis of the RespoKare® line of masks.
Innonix RESPOKARE RK-200-3040A RK-200-3041A RK-200-3042A N95 Mask is designed by Innonix, a company at the forefront of cutting-edge innovation in personal protective equipment for over many years, FDA registered.
Suggested applications of Respokare rk200 mask for General usage. The surgical function of the respokare NIOSH N95 mask helps nurses or doctors in a medical environment.
 Reliable
Reliable
Respokare N95 offers more than at least 95% filtration efficiency against certain non-oil-based particles, tested and approved for Respokare NIOSH n95.
The first US FDA-cleared mask to destroy 99.99% of human coronavirus within minutes, in America’s COVID-19 Hotspot.
Nelson Labs tested and approved according to 42 CFR Part 84
 Special anti-viral function
Special anti-viral function
As Respokare antiviral mask, it can kill 99% of bacteria and viruses on its surface in moments. Special Respokare materials layer with metal ions have an electrochemical effect, effectively killing the micro virus.
 Surgical function
Surgical function
FDA(510K) approved surgical function for Respokare RK200 N95 mask.
 Humanized Design
Humanized Design
3 sizes of respokare masks for an option which can fit most of the facial sizes.
Adjustable nose clip and elastic headband.
Specifications
| Brand | RESPOKARE |
|---|---|
| Model | RK-200-3040A RK-200-3041A RK-200-3042A |
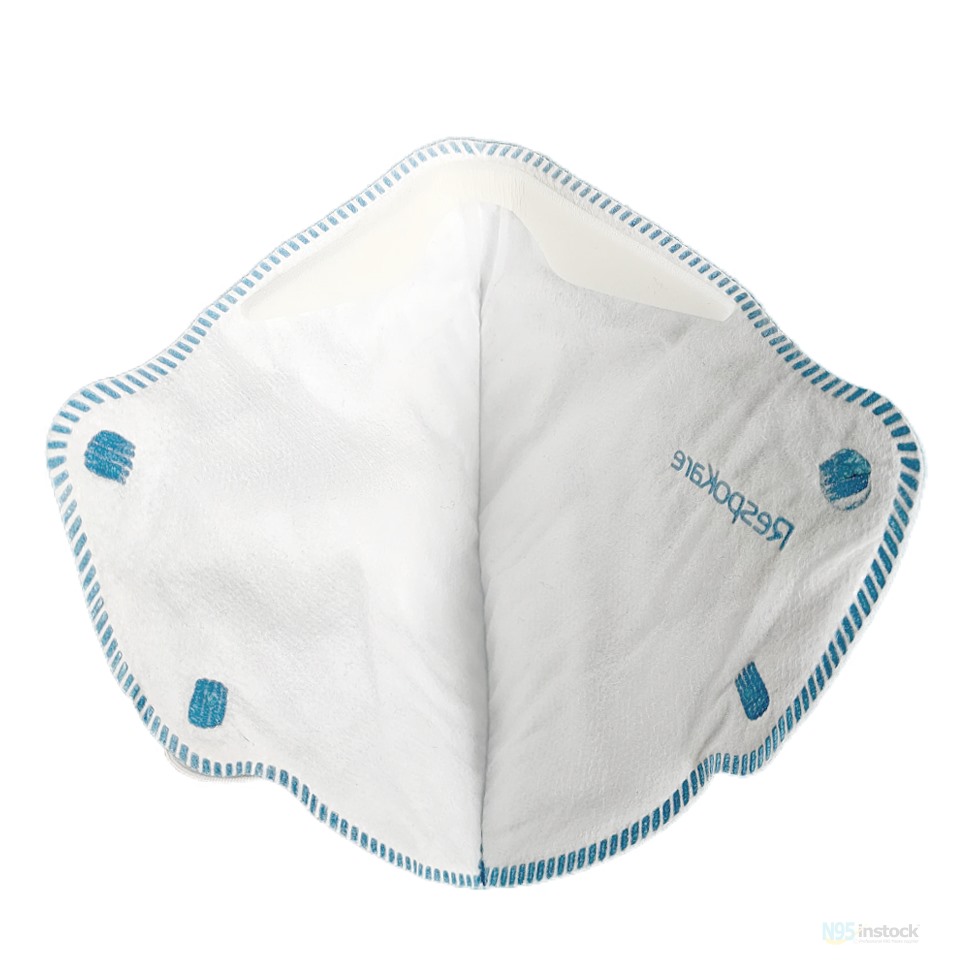
| N95 TC Number | TC-84A-7796 |
| Approval Date | 3/10/2017 |
| Manufacturer | Innonix Technologies Limited |
| Style | Headstrap |
| Shape | Folding |
| Strap Attachment Type | Welded |
| Latex | Free |
| Nose Clip | Metal |
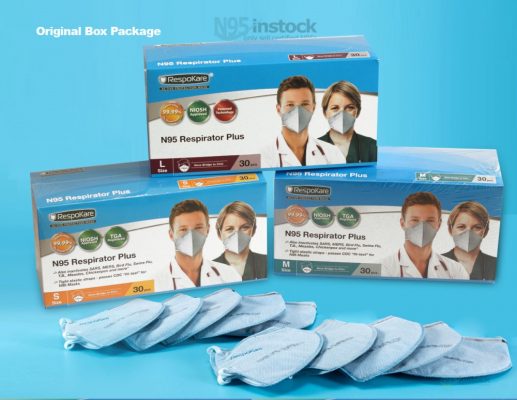
| Quantity Per Box | 30pcs |
| Shelf Life | 3Years |
 Features
Features
- 99. 99% inactivation of 15 different flu strains within 5 minutes.
- Be effective against 18 types of Flu Viruses, surrogates of SARS, MERS, and other pathogens.
- Adjustable nose clip and elastic headband help provide a custom secure seal.
- Flat fold design provides convenient storage and portability.
- 3 sizes for an option that can fit for most of the facial sizes.
- CDC NIOSH-approved masks.
- At least 95% filtration efficiency.
Application
Agricultural Production, Cleaning, Construction, Food and Beverage, Manufacturing, Grinding, Textile Operations
Industry
Construction, Food Processing, General Manufacturing, Industrial Maintenance, Transportation, Health Care
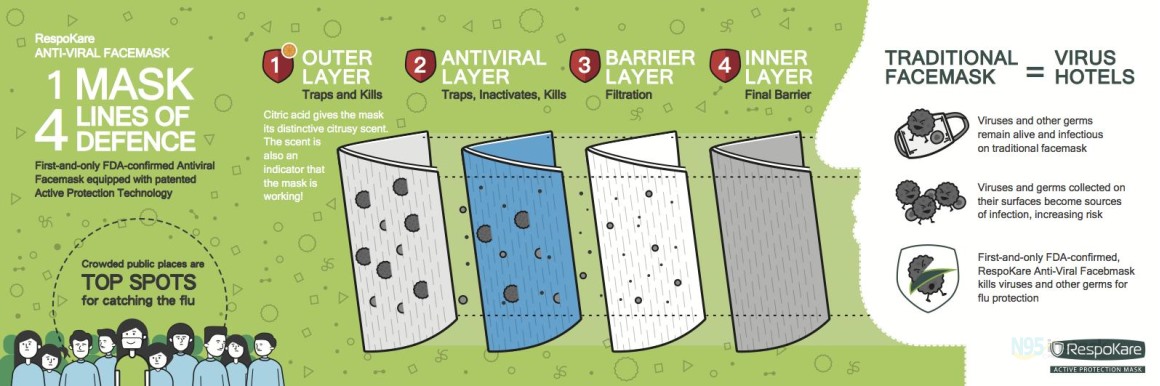
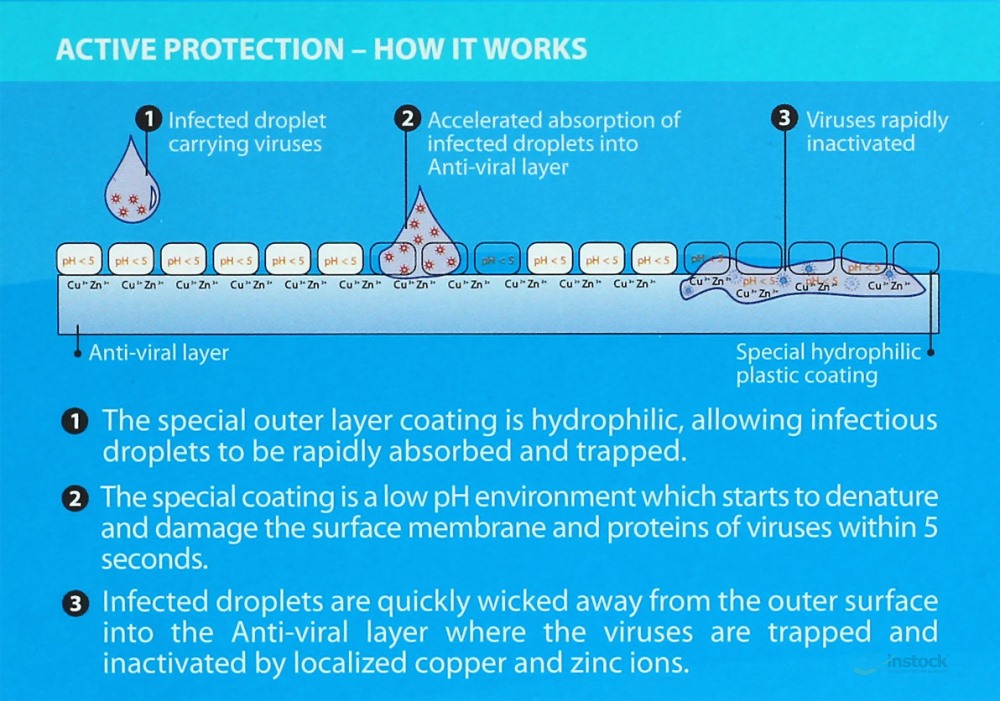
Anti-Viral Performance on Viruses Including Corona Viruses
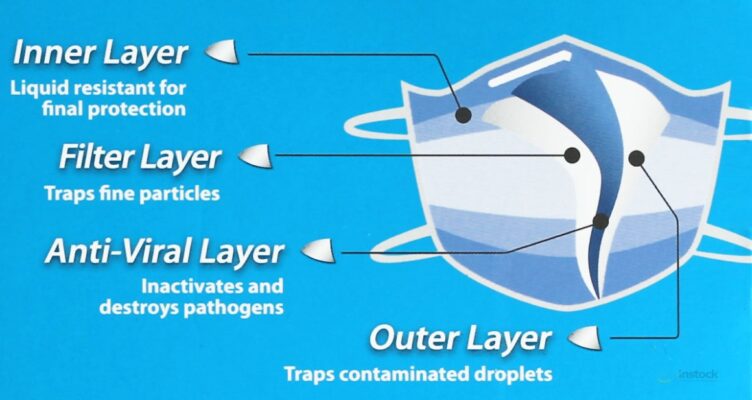
Respokare materials on the anti-viral layer
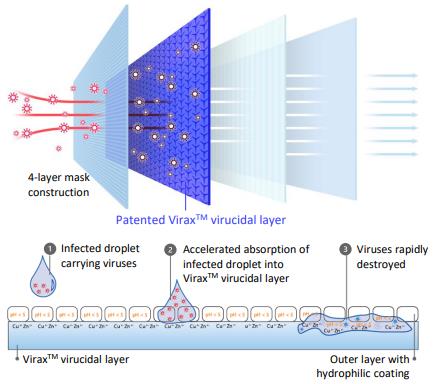
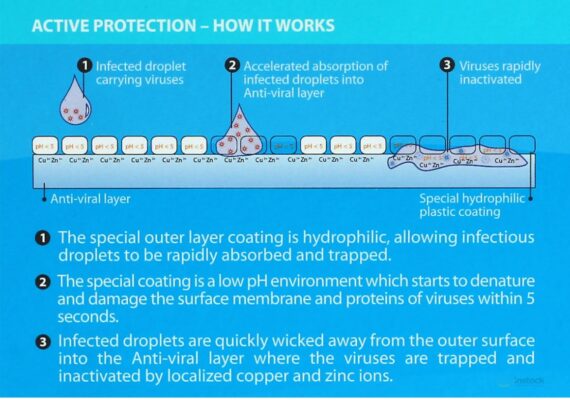
The outer layer coating is hydrophilic, allowing infected droplets to be rapidly absorbed and trapped. The coating’s low pH environment starts to denature and damage the surface membrane of viruses within 5 seconds. Infected droplets are quickly wicked away from the outer surface into the ViraxTM virucidal layer where the viruses are trapped and destroyed.
Anti-viral Layer – Biotechnology
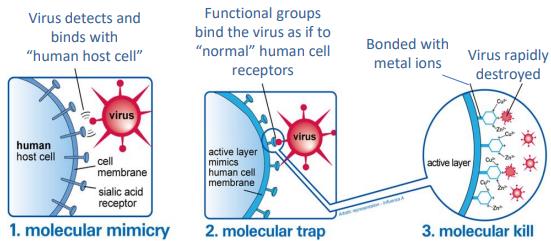 •The Anti-viral layer is a rayon material treated with active
•The Anti-viral layer is a rayon material treated with active
functional groups that mimic sialic acid found on the surface of
human cells.
•Viruses trapped in this layer are exposed to small but highly
localized concentrations of copper and zinc ions which destroy the
viruses through interaction with the viruses’ proteins, RNA or DNA.
Respokare anti-viral capability of RK-200 inactivates:
SARS and MERS (Coronaviruses):
99.99% inactivation of standard surrogate coronavirus within 1 minute
Measles, airborne transmission:
99.99% inactivation within 5 minutes
Tuberculosis, one of the deadliest bacteria causing 10 million ill and 1.6 million deaths each year:
88.97% inactivation within 10 minutes
The Anti-Viral Respirator also inactivates 99.99% of influenza Viruses:
99.99% inactivation of 15 different flu strains within 5 minutes

 Instruction
Instruction
99. 99% inactivation of 15 different flu strains within 5 minutes. highly effective against 18 common Seasonal & Pandemic Influenza. Adjustable noseclip helps provide a custom secure seal. The antimicrobial and/or antiviral agent kills specified pathogens under specified contact conditions. Flat fold design provides convenient storage and portability.
 WARNING
WARNING
- This mask does not eliminate the risk of contracting any disease or infection.
- Replace if damaged or soiled with blood or other bodily fluids.
- Do not use if hypersensitive to copper or zinc.
- If the mask becomes soiled please change to a new mask as the anti-microbial agents may not be effective.
- This mask does not contain natural rubber latex.
- For adult use only.
Authoritative reference of Respokare N95 RK200
 Publisher: NIOSH and FDA
Publisher: NIOSH and FDA
 Kevin MacDonald, Managing Director of Innonix Technologies USA LLC
Kevin MacDonald, Managing Director of Innonix Technologies USA LLC
“Innonix Technologies is a global biomedical materials R&D company with numerous intellectual properties and patents developed by American scientists. We are creating awareness of our premium masks and offering them to people who are looking for a quality mask with added anti-viral technology. We hope to play a role in mitigating Arizona’s pandemic. To date, thousands of masks have been donated to the Phoenix and Glendale Police Departments. Plans are in the works to donate several thousands of masks to in-classroom teachers and low-income senior living centers ”
 Certification
Certification
NIOSH Approved
Innonix RESPOKARE RK-200-3040A/3041A/3042A N95 NIOSH Approval Number TC-84A-7796, approval date 3/10/2017.
FDA cleared (510K)
Innonix RESPOKARE RK-200-3040A/3041A/3042A N95 mask passed FDA (510K) Cleared, worthy of consumer
Respokare rk200 n95 Product photo album
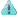 Cautions and Limitations
Cautions and Limitations
General Cautions and Limitations for All Part 84A Approvals Non-powered Air-Purifying Particulate Filter Respirators. These limitations are by no means all-inclusive.
FIT-CHECK THE FACE-SEAL AS FOLLOWS:
. Place both hands over the respirator do not disturb the position of the respirator.
. Inhale sharply (a negative pressure should be felt inside the respirator).
. If you detect air leaks, readjust the head straps and/or the nosepiece.
User Instruction
A. Not for use in atmospheres containing less than 19. 5% oxygen.
B. Not for use in atmospheres immediately dangerous to life or health.
C. Do not exceed maximum use concentrations established by regulatory standards.
D. Failure to properly use and maintain this product could result in injury or death.
E. All approved respirators shall be selected, fitted, used, and maintained in accordance with MSHA, OSHA, and other applicable regulations.
N Never substitute, modify, add, or omit parts. Use only exact replacement parts in the configuration as specified by the manufacturer.
F. Refer to user’s instructions, and/or maintenance manuals for information on the use and maintenance of these respirators.
G. NIOSH does not evaluate respirators for use as surgical masks.
H. Special or Critical User’s Instructions: Before occupation use of this respirator a written respiratory protection program must be implemented meeting all the local government requirements. In the United States, employers must comply with OSHA 29 CFR 1910. 134 which includes medical evaluation, training, and fit testing.
What they're saying
Very comfortable and safe for my daily use. Although the price is higher than other masks I have used, it is still worth using.
Respokare n95 masks have three sizes, size small and medium are fit, my family. The color also looks cool. The size large isn’t fit me. So I gave the old one to my friend and buy a new one in size medium.
Initially, it has a special smell but is acceptable; I still send an email to Instocking customer service asking for a reason. Their customer service explained that the smell from the outer layer is acidic which was coated with a special hydrophilic plastic, aiming to inactivate the virus. They guided me to open the box to dissipate the smell for some days. I followed and it do better.












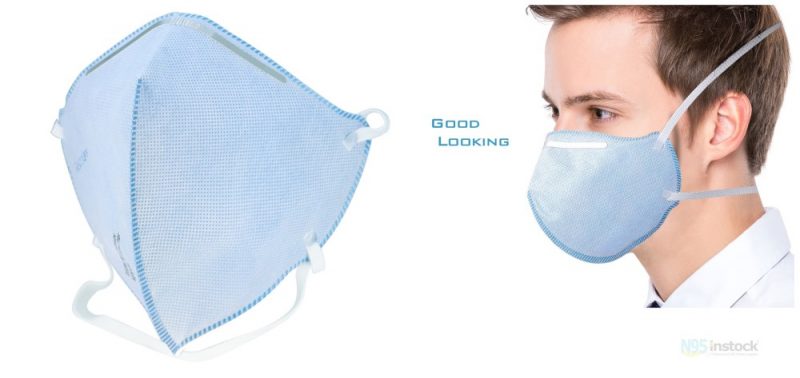
































This RESPOKARE N95 mask is a genuine, authorized product. The anti-viral technology offers an extra layer of defense.
I love how it professional grade protection. I love how it high filtration efficiency.
The RESPOKARE RK200 series offers sizes for everyone (I got small 3040A). The anti-viral tech to kill viruses on the surface is brilliant.
These masks are great and well-made. I appreciate that they provide authorized distributor while still RESPOKARE N95 brand. Excellent value for money. The quality is noticeably better than similar products I’ve tried. The RESPOKARE N95 brand is particularly impressive. I’ve already recommended it to several friends and colleagues.
Very happy with the RESPOKARE N95. The anti-viral technology is a significant benefit. Comes in different sizes for a good fit.
I would definitely recommend this to others. Great purchase! I love how it breathable yet protective.
As a frequent traveler, I’m impressed with the hot selling of these masks. RESPOKARE N95 brand makes them perfect for regular wear. Perfect balance of quality and price. It’s exactly what I was looking for in a mask product.
I love how it secure fit. This product is reliable. This product is reliable.
I chose the RESPOKARE N95 RK200 (medium size 3041A) for its virus-killing technology. Gives me extra peace of mind.
As someone who needs masks for professional settings, I find these outstanding and sturdy. The 30 pieces per box is exceptional. Exceeds expectations. The 30 pieces per box is particularly impressive. I’ve already recommended it to several friends and colleagues.
These masks are perfect for travel due to their RESPOKARE N95 brand. The hot selling is impressive compared to other brands. This will be my go-to product from now on.
I’ve tried many masks, but this one stands out because of its authorized genuine. hot selling is a huge plus. I appreciate the attention to detail.
The RESPOKARE N95 mask with anti-viral layer is a fantastic product. Knowing it kills viruses on the surface is very reassuring.
This product is premium. I love how it professional grade protection.
This product is wonderful. This product is comfortable. I love how it secure fit.
As someone who needs masks for daily use, I find these wonderful and great. The 30 pieces per box is exceptional. Very satisfied with this purchase.
This product is top-notch. I love how it comfortable for all-day wear. This product is reliable.
This RESPOKARE RK200 mask is a game-changer. Anti-viral, NIOSH approved, FDA cleared ¨C it has all the protection I need.
I love how it excellent filtration. I love how it comfortable for all-day wear.
This RESPOKARE N95 mask is innovative! The anti-viral feature is a huge plus on top of NIOSH and FDA clearance. Got the large size (3042A).
These masks are well-made and effective. I appreciate that they provide RESPOKARE N95 brand while still authorized genuine. The quality is consistent and reliable.
These masks are comfortable and convenient. I appreciate that they provide 30 pieces per box while still niosh approved. Perfect balance of quality and price.
I would definitely recommend this to others. Great purchase! This product is perfect.
The RESPOKARE N95 Mask RK200 series is amazing with its anti-viral technology! NIOSH approved and FDA cleared, perfect for protecting my family.
reasonal price, genuine, shipment could faster
reasonal price, genuine, good service
These masks are efficient and reliable. I appreciate that they provide hot selling while still niosh approved. Excellent value for money. I’ve been using it for 5 months now with consistent results.
I would definitely recommend this to others. Great purchase! I love how it perfect seal around the face. I love how it excellent filtration.
This product is superb. This product is durable.
The RESPOKARE N95 RK200 series is a hot-selling item for a reason. The anti-viral protection is a standout feature.