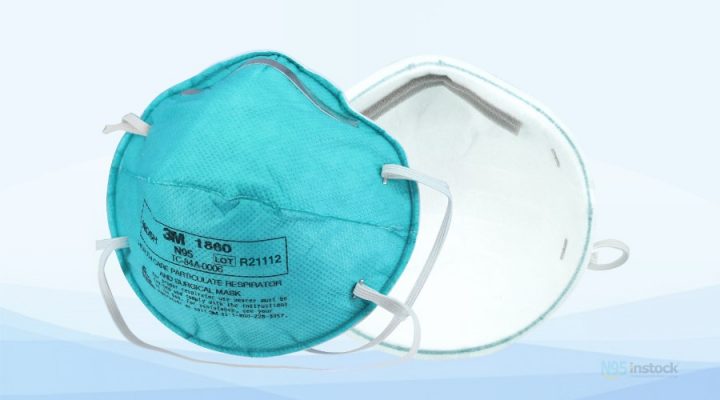
3M 1860 N95 Mask details
3M 1860 N95 surgical mask contains no components made from natural rubber latex. It meets CDC guidelines for Mycobacterium tuberculosis exposure control. As a surgical mask, it is fluid resistant to splash and spatter of blood and other infectious materials. It also provides >99% BFE2 against wearer-generated micro-organisms. it complies with the OSHA Bloodborne Pathogens Standard. Tested against particles approximately 0. 3-micron particle (mass median aerodynamic diameter) per US 42 CFR 84. Bacterial Filtration Efficiency (BFE) is determined by the ASTM F2101 test method. Fluid resistant according to ASTM F1862.
Specifications
| Brand | 3M |
|---|---|
| Model | 1860 |
| N95 TC Number | TC-84A-0006 |
| Approval Date | 8/17/1995 |
| Manufacturer | 3M Company |
| Style | Headstrap |
| Shape | Cup |
| Strap Attachment Type | Braided Comfort Strap |
| Latex | Free |
| Nose Clip | Metal |
| Quantity Per Box | 20pcs |
| Shelf Life | 5 Years |
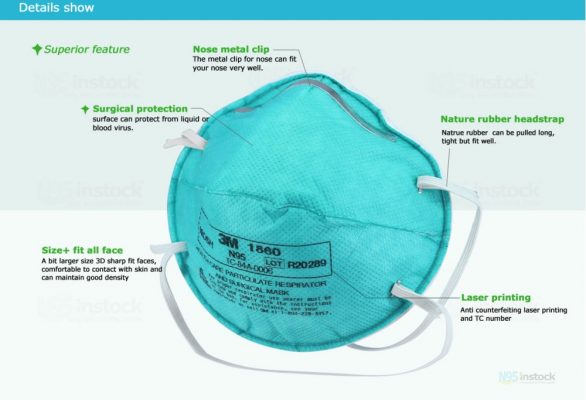
Features
- Surgical N95 mask for use in healthcare.
- Filter efficiency of 95% or greater against particulate aerosols free of oil.
- Provides >99% BFE against wearer-generated micro-organisms according to ASTM F2101.
- Be designed to be fluid resistant to splash and spatter of blood and other infectious materials.
- Complies with the OSHA Blood-borne Pathogens Standard.
- Braided headbands for various facial sizes
- Cushioning nose foam, and lightweight construction for comfortable wear.
Application
Agricultural Production, Airborne Biological Particles, Bagging, Dry Chemical Handling, Emergency & Pandemic Preparedness, Foundry Operations, Grinding, Petrochemical Manufacturing, Processing of Minerals, Sanding, Textile Operations
Industry
Operating Rooms, Clinics, TB Wards, Patient Care, Labor and Delivery, Infection Control Practices, Laboratory, emergency or pandemic preparedness planning, stockpiling, etc.
Instruction
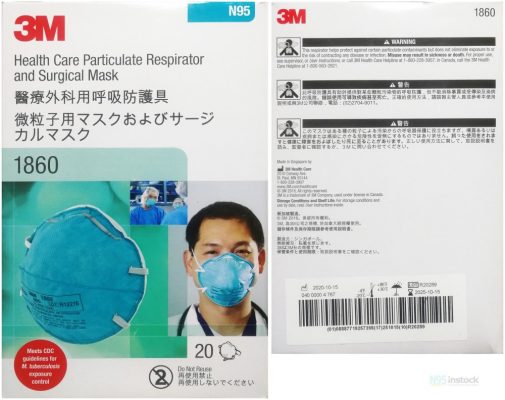
The 3M 1860 N95 Mask Health Care Particulate Respirator and Surgical Mask are designed to help provide respiratory protection for the wearer. 3M 1860 N95 Mask is NIOSH approved and FDA 510k cleared. FDA Cleared it for use as a surgical mask. This product has a filter efficiency level of 95% or greater against particulate aerosols free of oil1. It is fluid-resistant, disposable, and may be worn in surgery. It can fit a wide range of face sizes. This product meets CDC guidelines for Mycobacterium tuberculosis exposure control. As a respirator, it is intended to reduce wearer exposure to certain airborne particles in a size range of 0.1to >10.0 microns, including those generated by electrocautery, laser surgery, and other powered medical instruments.
WARNING
These respirators help reduce exposures to certain airborne contaminants. Before use, the wearer must read and understand the User Instructions provided as a part of the product packaging. In the U.S., a written respiratory protection program must be implemented meeting all the requirements of OSHA 1910.134 including training, fit testing, and medical evaluation.
Do Not Use For
- Medical N95 mask is not for industrial use.
- Not for use with beards or other facial hair that prevents direct contact between the face and the sealing surface of the respirator.
- OSHA has not set a permissible exposure level for airborne biohazards.
Certification
NIOSH Approved
3M 1860 N95 NIOSH Approval Number TC-84A-0006, approval date 8/17/1995
FDA cleared (510K)
3M 1860 passed FDA (510K) Cleared, worthy of consumer
3M 1860 N95 face mask photo album
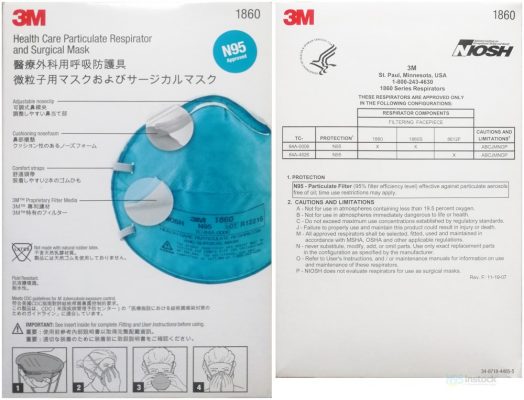
Cautions and Limitations
General Cautions and Limitations for All Part 84A Approvals Non-powered Air-Purifying Particulate Filter Respirators. These limitations are by no means all-inclusive.
FIT-CHECK THE FACE-SEAL AS FOLLOWS:
. Place both hands over the respirator do not disturb the position of the respirator.
. Inhale sharply (a negative pressure should be felt inside the respirator).
. If you detect air-leaks, readjust the head straps and/or the nose-piece.
User Instruction
- The 3M saccharin (sweet) or Bitrix (bitter) qualitative fittest is recommended for this respirator. When used only as a surgical mask, fit testing is not required.
- A respirator may be used until damaged, breathing becomes difficult, or contaminated with blood or body fluids. Otherwise, it may be stored and reused according to the facility’s infection control policy.
- Filtering face pieces are to be inspected prior to each use to assure there are no holes in the breathing zone other than the punctures around staples, and no damage has occurred. Enlarged holes resulting from ripped or torn filter material around staple punctures are considered damage. Immediately replace the respirator if damaged. Staple perforations do not affect NIOSH supplier approval.
- Discard after every use when used for surgical procedures.
- Not for atmospheres containing less than 19. 5 percent oxygen.
- Not for atmospheres immediately dangerous to life or health.
- Do not exceed maximum-use concentrations established by regulatory standards.
- Failure to properly use and maintain this product could cause injury or death.
- This respirator is designed for occupational/professional use by adults who are properly trained in their use and limitations. Respirators are not designed to be used by children.
What they're saying
This is a great mask I use it to protect myself I always bought this one because of the extra protection. Doesn’t t fog my glasses. Better than any mask I’ ve had so far.
A week later I got the mask package and compared it with the 3M 1860 I had. It’s genuine.
They were two days later than expected, but they were the real deal. The printing on the mask is very clear.



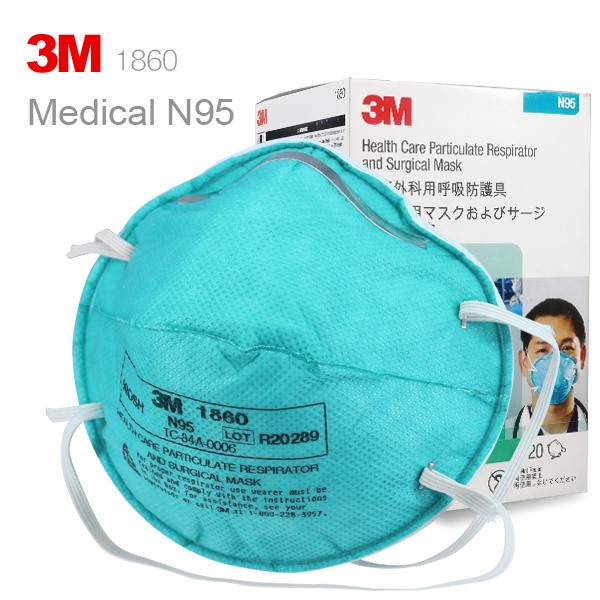


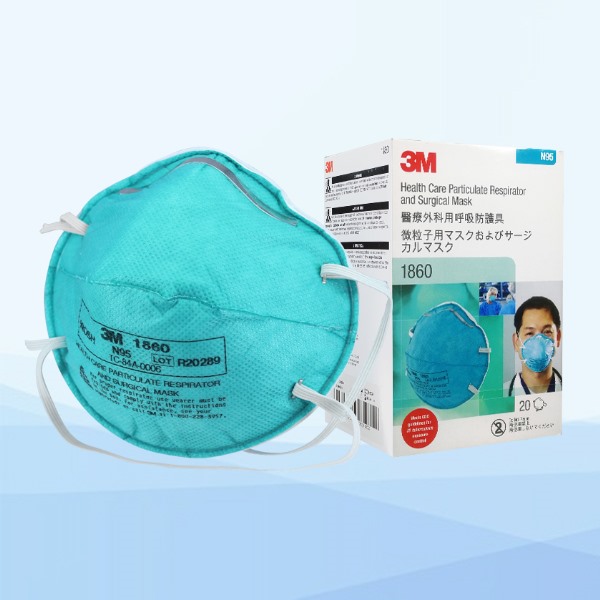














































I would definitely recommend this to others. Very satisfied! I love how it easy to breathe through. This product is fantastic.
I’ve tried many masks, but this one stands out because of its comfortable fit. N95 Mask brand is a huge plus. This will be my go-to product from now on. I’ve been using it for 2 months now with consistent results. The comfortable fit is particularly impressive.
This product is perfect. I love how it easy to breathe through.
I love how it doesn’t fog up my glasses.
These masks are top-notch and amazing. I appreciate that they provide comfortable fit while still niosh approved. Best product in its category. It’s become an essential part of my daily routine. I’ve already recommended it to several friends and colleagues.
These masks are durable and durable. I appreciate that they provide comfortable fit while still niosh approved. I appreciate the attention to detail.
I would definitely recommend this to others. Very satisfied! I love how it professional grade protection.
As a retail worker, I’m impressed with the secure seal of these masks. easy breathing makes them perfect for daily use. Perfect balance of quality and price.
This product is sturdy.
These masks are perfect for outdoor activities due to their secure seal. The skin-friendly material is impressive compared to other brands. The quality is consistent and reliable.
These masks are convenient and fantastic. I appreciate that they provide secure seal while still niosh approved. The quality is consistent and reliable.
I would definitely recommend this to others. Very satisfied! This product is fantastic. I love how it excellent filtration.
As a office worker, I’m impressed with the secure seal of these masks. N95 Mask brand makes them perfect for regular wear. Very satisfied with this purchase. It’s exactly what I was looking for in a mask product.
As someone who needs masks for office environments, I find these sturdy and high-quality. The easy breathing is exceptional. Will definitely purchase again. The N95 Mask brand is particularly impressive.
This product is high-quality. This product is fantastic. This product is efficient.
I’ve tried many masks, but this one stands out because of its comfortable fit. skin-friendly material is a huge plus. Exceeds expectations.
These masks are perfect for professional settings due to their niosh approved. The secure seal is impressive compared to other brands. Will definitely purchase again. The quality is noticeably better than similar products I’ve tried. It arrived well-packaged and in perfect condition.
I love how it high filtration efficiency. I love how it doesn’t fog up my glasses. This product is convenient. I love how it breathable yet protective.
I’ve tried many masks, but this one stands out because of its skin-friendly material. niosh approved is a huge plus. This will be my go-to product from now on.
As someone who needs masks for regular wear, I find these superb and efficient. The easy breathing is exceptional. Highly recommended!
As a office worker, I’m impressed with the secure seal of these masks. N95 Mask brand makes them perfect for office environments. Perfect balance of quality and price. It’s become an essential part of my daily routine. It arrived well-packaged and in perfect condition.
As someone who needs masks for regular wear, I find these sturdy and durable. The secure seal is exceptional. Very satisfied with this purchase. It’s become an essential part of my daily routine. The secure seal is particularly impressive.
This product is excellent.
I love how it high filtration efficiency.
I love how it easy to breathe through. This product is great. I love how it comfortable for all-day wear.
These masks are sturdy and durable. I appreciate that they provide niosh approved while still 20 pieces per box. Excellent value for money.
This product is reliable. I would definitely recommend this to others. Very satisfied! This product is comfortable. I love how it perfect seal around the face.
These masks are perfect for travel due to their 20 pieces per box. The niosh approved is impressive compared to other brands. Highly recommended! The quality is noticeably better than similar products I’ve tried. It’s become an essential part of my daily routine. I’ve already recommended it to several friends and colleagues.
These masks are durable and durable. I appreciate that they provide skin-friendly material while still N95 Mask brand. Perfect balance of quality and price.
These masks are perfect for office environments due to their comfortable fit. The 20 pieces per box is impressive compared to other brands. Excellent value for money.