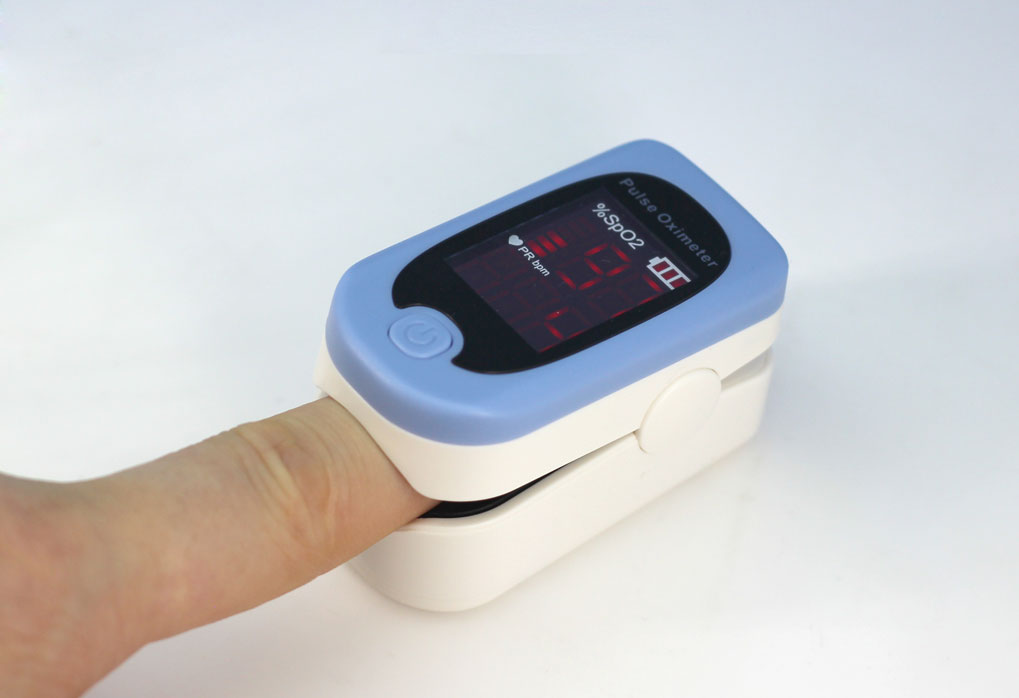
Fingertip Pulse Oximeter details
ACCARE FS10D Blood Oxygen Saturation Monitor measures blood oxygen saturation and pulse rate. It is manufactured by the Hunan Accurate Bio-Medical Technology Company, a well-regarded leader in fingertip pulse oximetry. The device is FDA registered. Meanwhile, it is also 510K approved with number K141105. Simple operation allows it to turn and off with the touch of a button. And you can expect up to 600 spot checks on a single set of fresh batteries.
Specifications
| Brand | Accare |
|---|---|
| Model | FS10D |
| Certification | CE/ FDA/ 510K |
| Manufacturer | ACCURATE BIO-MEDICAL TECHNOLOGY |
| Battery | 2* AAA BATTERIES |
| Package Size | 3.1 x 2.2 x 2.2 in |
| Item Weight | 2.12 oz |
Features
- Easy to use with one button touch.
- Accurate, fast, and reliable Fingertip Pulse Oximeter accurately determines your SpO2 in less than 10 seconds.
- Large and easy-to-read multi-view LED display screen.
- Wireless and be portable with lightweight.
- Low in power consumption.
- 3 models available: power off, sleep, and measure.
- Automatically turning into sleep mode with 8 seconds if no signal.
- Accommodates a range of finger sizes from pediatric to adult.
Application
Sports & Aviation use only
Industry
Sports
Instruction
As a basic type fingertip pulse oximeter, the FS10D has a bright LED display. Therefore, it is easy to read. We intend the device for adult and pediatric usage, but not for infants or newborns. We intend it for use by clinical professionals or under their guidance. It has a low power consumption in that 600 spot checks can be made with the supplied 2 AA batteries. To further conserve battery life, the unit goes into sleep mode if not used within 8 seconds of being turned on.
WARNING
- We intend this device for Sports & Aviation use only, and it is not a medical device.
- Like almost all finger pulse oximeters, this unit will NOT monitor SPO2 during activity and movement. You must stop the current activity and take the reading while stationary. The movement will cause artifacts to affect readings.
Certification
CE
FDA
510K
Cautions and Limitations
- Do not share with NMR or CT equipment.
- Do not use this monitor in a flammable anesthetic gas environment.
- Not intended to diagnose, treat, cure, or prevent any disease or health condition.
- Please carefully read all labeling and instruction menus before use.
Below factors may cause incorrect readings
- Bright nails or nails with nail polish.
- Immersion of sensors in liquid disinfectant.
- Intravascular staining.
- Accidental movements of the user.
- The interference of medical high-frequency signal.















Reviews
There are no reviews yet.